1. Specifications
Fig.1 illustrates the structure of a typical capillary column. Both the inlet frit and the outlet frit are sintered in the column with the packing materials. The detection window is created about 2mm after the outlet frit by carefully removing the polyimide coating of the capillary. ElectropakTM columns are available in 50mm, 75mm, 100mm and 320µm i.d. capillaries packed with 1.5mm to 7mm porous or non-porous HPLC packing materials. Many stationary phases including C18, C8, phenyl, cyano, silica, ion exchanger and size exclusion are available. The length of the packed bed may be selected according to the CE or CEC instrument used (e.g., 20cm or 30cm for Beckman’s P/ACE and 25cm or 40cm for) Hewlett-Packard’s HP3DCE—with lengths typically ranging from 20 cm to 50 cm.
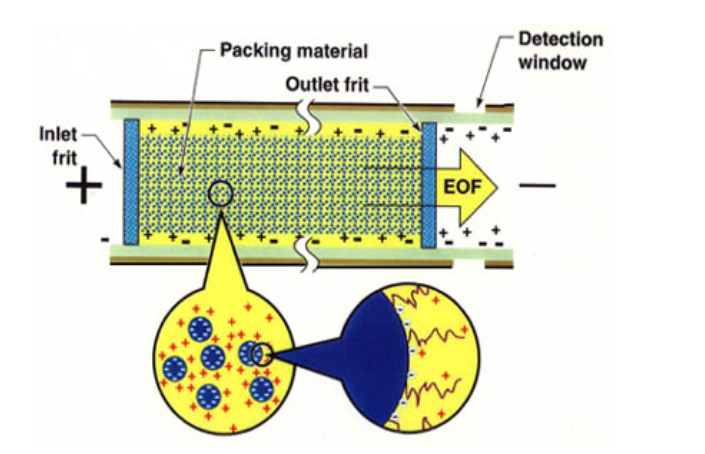
Fig. 1 Structure of a packed capillary column
Capillary column stationary phase and its applications:
| Stationary Phase Name |
Bounded Funtional Group |
Properities |
Chromatographic Separation Mode |
Application |
| C8 |
-C8H17 |
Non-polar |
Reversed-phase, ion-pair |
Medium-polarity compounds, highly polar water-soluble compounds such as: small peptides, proteins, steroids, nucleobases, nucleosides, nucleotides, polar synthetic drugs, etc. |
| C18 |
-C18H37 |
|
|
|
| Phenyl |
-C6H5 |
Non-polar |
Reversed-phase, ion-pair |
Non-polar to medium-polarity compounds, such as fatty acids, glycerides, polycyclic aromatic hydrocarbons, esters (phthalates), fat-soluble vitamins, steroids, PTH-derivatized amino acids. |
| Amino |
-(CH₂)₃NH₂ |
Polar |
Normal-phase, reversed-phase, anion-exchange |
Normal-phase: Separates polar compounds such as aromatic amine derivatives, lipids, steroids, chlorinated pesticides.
Reversed-phase: Separates monosaccharides, disaccharides, polysaccharides and other carbohydrates. Anion-exchange: Separates phenols, organic carboxylic acids and nucleotides.
|
| Cyano |
-(CH₂)₃CN |
Polar |
Normal-phase, reversed-phase |
Normal-phase: Similar to silica gel, acting as a hydrogen bond acceptor, suitable for analyzing polar compounds with lower solute retention than silica columns. Reversed-phase: Provides different selectivity compared to C8, C18, and phenyl columns. |
| Strong Cation Exchange |
-(CH₂)₃C₆H₈SO₃H⁻ |
|
Strong cation exchange |
Organic bases |
| Strong Anion Exchange |
-(CH₂)₃N⁺(CH₃)₃ |
|
Strong Anion Exchange |
Organic acids, nucleosides and nucleotides |
| Silica |
-OH |
Polar |
Normal-phase |
Non-polar, medium-polarity and non-ionic organic compounds |
Other details:
- Transportation:By air if not specified.

